


The EVOL® SI features a super hydrophilic HAnano Surface that has proven to accelerate bone growth in over 30 in vivo and in vitro studies.* Faster and stronger osseointegration has been demonstrated through biomechanical, histomorphological, and biological evaluations, in combination with over 150,000 clinical applications to date.
This technology improves osseointegration through the combination of high wettability and optimal surface chemistry (with optimized nano-roughness) that mediates bioactivity and specific protein adsorption to the implant. The nanothin surface enables newly formed bone to grow directly into the micrometer topography, or roughness, of the implant surface, thus providing mechanical stability with no risk of cracking or delamination of the implant.System features:
- 20 nm Thin HA Surface Treatment
- Crystalline HA, through size, shape and structure, mimics living bone tissue
- Eliminates concerns of delamination
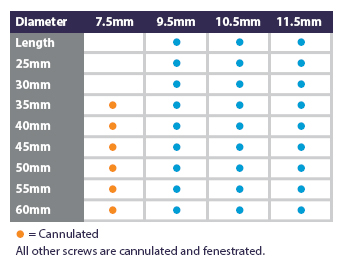
- Open Body Cannulation
- Provides ability to use guide wire for more accurate screw placement
- Integrated funnel allows streamlines graft packing post implant
- Fenestrated/Slotted Screws
- Multiple off-setting slots allow greater opportunity for fusion and SI Joint alignment
- Optional Polyaxial Washer
- Distributes load throughout a larger area of the pelvis
- Removal of washer affords placement flexibility
- Dual Thread Regions
- Cortical thread for purchase in ilium
- Cancellous thread for purchase in sacrum

HAnano Surface is a 20 nanometer thin implant coating composed of crystalline hydroxyapatite (HA) particles that through shape, composition, and structure mimic human bone tissue. HAnano Surface is used clinically and has proven to significantly accelerate and enhance osseointegration of implants. The surface can be used on all implants regardless of material and geometry.
*Jimbo et al. (2012), ‘The biological response to three different nanostructures applied on smooth implant surfaces’, Clinical Oral Implants Research, vol. 23, no 6, pp. 706-712
For additional information, please contact:
Phone: 877-780-4370 Fax: 601-420-5501

